
Complication profile of transanal total mesorectal excision and how it differs
Introduction
Surgery for rectal cancer has drastically evolved over the last four decades. Transanal total mesorectal excision (taTME) has organically developed over the last decade to improve access to tumors of the mid and distal rectum with a minimally invasive approach, especially in obese patients or those with a narrow pelvis. Despite encouraging data on outcomes (1,2), it still remains a controversial procedure due to its risks of complications and the potential for increased risk of local recurrence. Unique changes in technique and surgical anatomy are required to successfully perform taTME, in addition to a change in instrumentation. Proper insertion and setup of transanal access platforms and safe utilization of newer continuous flow insufflators is mandatory. The effects of pneumodissection on surgical planes, coupled with unfamiliar anatomy, increases the risks of adjacent organ and structure injury, highlighted by the development of the most feared complication of taTME surgery, urethral injury. More recently, the increasing incidence of gas embolism has been elucidated, striking further fear and concern by surgeons. Potential increase in anastomotic failure, functional compromise through pelvic nerve injury, and the more traditional risk of surgical bleeding, are all at the forefront of concern. Despite the high complication rate of rectal resection performed by any technique of 20–35%, surgeons should be thoroughly educated and aware of the unique mechanism of complications in taTME, and how to avoid them. Simulation and proctored courses should be utilized in order to combat the steep learning curve and minimize complications of this surgical technique.
Pursestring failure
Placement of a circumferential submucosal pursestring below the level of the tumor is the first and most critical step in performing taTME, yet it is also the most challenging to perform. Precise endoluminal suturing is an uncommon skill set with great disparity amongst colorectal-trained surgeons. Placement of the initial pursestring is key, as it is required to maintain pneumorectum, it prevents fecal spillage into the field, and it ensures an adequate distal margin. Additionally, a secure pursestring is critical for pneumodynamics, as inadvertent insufflation of the proximal colon will make the abdominal dissection more challenging and minimize the effects of pneumodissection and working space for the transanal operator. Local recurrence likely due to tumor seeding after rectal perforation during abdominoperineal resections has been described (3). Thus, a similar outcome could occur if the pursestring is not secured completely and the surgical field is contaminated with fecal or tumor matter. A bactericidal washout with betadine should be performed after securing the pursestring.
Pursestring construction is critical. Using electrocautery to mark the proposed pursestring avoids spiraling, which leads to an insecure or asymmetric closure. After securing the pursestring knot in an “open” fashion, with the transanal cap off to ensure tightness, the closure should be checked by attempting to pass forceps through the center of the pursestring prior to insufflation (Figures 1,2). Recognizing a defect at this point can be quickly salvaged by redoing the pursestring or more easily by placing a figure of eight suture.
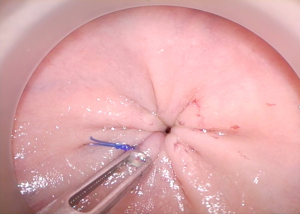
Recognition of an incomplete pursestring once proctotomy has been performed should be remedied by immediate closure and repeat washout. Easier rescue of the pursestring can be performed with an endoloop. Pursestring failure may be a result of excessive manipulation of the suture or insufficient suture depths with easy avulsion from the mucosa. Conversely, sutures that are placed too deeply may enter the endopelvic fascia posterolaterally or even pierce the prostatic fascia and vagina anteriorly. It is essential that the surgeon is satisfied with the quality of the pursestring prior to proceeding the remainder of the procedure in order to prevent potential catastrophe later.
Wrong plane dissection
As with any other aspect of colorectal surgery, wrong plane surgery often leads to injury to surrounding organs and neurovascular structures. Identification of the multiple planes of the rectal wall and pelvis following proctotomy, exacerbated by the effects of pneumopelvis, is unfamiliar to many surgeons. Careful, repetitive video observation, supportive supervision, proctorship, and the following recommendations all facilitate technique mastery and safety.
After a full thickness proctotomy, TME dissection should be done in an orderly, stepwise fashion. It is imperative that dissection is maintained circumferentially, to prevent significant distortion of the anatomy, as the rectum is forced by pneumopelvis towards the side of least dissection. As in nearly all abdominopelvic surgery, encountering bare muscle at any point is an indication that the plane is too deep. Even in the skinniest of patients, a thin layer of endopelvic fascia will overlay the pelvic floor or muscles of the pelvic side wall.
Through continued case and video observation, review of the taTME database, cadaveric training, and Delphi studies, several key areas of wrong plane surgery have been identified. Unlike conventional TME where the pelvis narrows and carries the risk of “coning in” on the oncologic specimen, the transanal surgeon is more at risk for inadvertent lateral dissection as they ascend the pelvis. Although the endopelvic fascia overlying the levator ani is easily identified posterolaterally early in the operation, continuing to follow the endopelvic laterally, and failing to appreciate the curvature of the mesorectum, quickly leads the surgeon far lateral to the correct dissection plane (Figure 3). In early experiences, the obturator lymph node packet with distinctive appearing fat, was frequently exposed medially as one dissects along the obturator internus muscle. If one proceeds too laterally, distal branches of the internal iliac vein and artery can be exposed and injured before they course through Alcock’s canal. Within the obturator fat, the obturator nerve, artery and vein can be injured as they cross diagonally. More ominously, dissection can wander lateral to the inferior hypogastric plexus as well as the S4 nerve root, exposing these structures to injury despite corrective maneuvers. Interestingly, the ease of exposure of the lateral pelvic lymph node packet from a transanal approach has led to pioneering work being done in Japan into transanal lateral lymph node dissection (TaLLND).
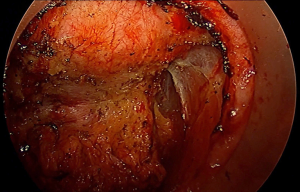
In early cadaveric work, posterior intramesorectal dissection was frequently encountered. The dramatic curvature of the lower sacrum and anorectum was underappreciated, and the ergonomics required for direct posterior dissection challenging. Early dissection in the midline is therefore discouraged, with initiation of dissection preferably posterolaterally (Figure 4). Furthermore, dissection between the rectal wall and mesocolon is erroneously encouraged by pneumodissection, easily creating the façade of the correct plane. Encountering the white appearing outside of the rectal wall as it curves posteriorly should immediately alarm the surgeon, in addition to perforating blood vessels between the mesocolon and rectal wall.
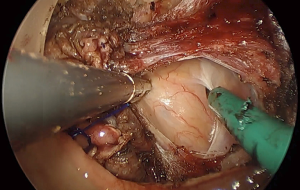
Finally, as one ascends the pelvis in the posterior plane, the sacrum abruptly curves. At the level of S3, Waldeyer’s fascia is incised to maintain the innermost plane to the mesorectum. Failure to follow the curvature leads to an incision through the presacral fascia and on to the exposed sacral vertebrae (Figure 5). The anatomy and pitfalls leading to urethral injury are separately described in detail.
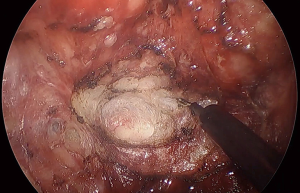
Vascular injury
Injuries to vascular structures with resultant bleeding are a risk with any surgical procedure and have been well described in traditional TME. The same vessels encountered in both the appropriate TME plane and inadvertent extramesorectal plane are similarly vulnerable during transanal TME. In general, the proximity to the vessel and angle at which it is approached from the transanal viewpoint permits easier identification and control. As a rule, the vessel should be handled from the same trajectory as the occurrence; thus, attempting to control from the top is more challenging with inferior exposure. As with the conventional approach, injury to the presacral plexus of veins deep to the presacral fascia is not uncommon and often occurs at the same location. Failure to follow the acute curvature of the sacrum during proximal dissection takes the surgeon deep to presacral fascia. Bleeding can be controlled by direct compression with a dry gauze or bipolar cautery easily from this orientation. Dissection directly on the mesorectal envelope helps to avoid this occurrence. The middle rectal arteries, described as coursing within the lateral stalks, vary in their origin, independence, and true presence. Anatomical studies demonstrate presence of bilateral middle rectal arteries as little as 12% of the time. When present, nearly 70% have a common origin, with prostatic arteries forming a large prostate-rectal trunk. During traditional TME, they are often handled with impunity, divided without clear visualization using advanced bipolar energy or, more commonly, with simple monopolar cautery. From the transanal approach, they seem to appear with increased clarity requiring definitive treatment. Middle rectal artery identification in the lateral stalks is easier via the transanal approach, thus allowing improved exposure for a transabdominal transection with bipolar energy if there are two teams working in tandem, as with synchronous taTME.
The remaining causes of vascular injuries generally occur from inappropriate extramesorectal dissection, usually from inadvertent lateral dissection where venous and arterial branches of the terminal internal iliac branches are found. Additionally, extramesorectal vasculature can be injured from tenting in during retraction, especially the anterior arteries supplying the urogenital organs which can be easily manipulated to assume a more perpendicular orientation (Figure 6).
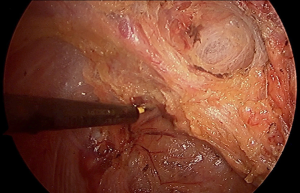
Pelvic nerve injury
The lower third of the rectum is innervated by a complex network of autonomic nerves composed of the superior hypogastric plexus made of sympathetic nerves, the inferior hypogastric plexus comprised of sympathetic and parasympathetic nerves, and the pelvic splanchnic nerves. During dissection of the infraperitoneal rectum, the autonomic nerves and plexuses are at high risk of injury, especially during dissection of the rectum from the prostate, seminal vesicles, and vagina. Nerve injury during total mesorectal excision, occurs most commonly from excessive lateral dissection, extramesorectal dissection, and as a retraction injury. Urologic and sexual outcomes have been studied extensively in open, laparoscopic, and robotic surgery with a wide range of reported dysfunction, 11–85%, and overall equivalence between techniques (4,5). However, the majority of cohort studies demonstrate strong selection bias, and furthermore, include learning curves during the adoption of minimally invasive surgery over the last two decades. A single randomized trial in a small cohort of distal third rectal cancers suggested superior outcomes of sexual function favoring the transanal approach (6).
TaTME leaves the autonomic plexus similarly vulnerable to nerve injury as previous techniques. Although enthusiasts and early adopters often cite improved visualization, superior access, and easier retraction as to why nerve injury may be less likely, the mechanics of extrarectal pneumodissection, combined with the difficulties of a novel anatomic approach and the requirement of advanced minimally invasive skills, may leave the nerves even more susceptible to damage, especially early in the learning curve. A thorough understanding of the nerve distribution seen from the transanal approach, combined with precise mesorectal dissection, is critical to avoid unnecessary nerve injury and resultant impaired functional outcomes from poor surgical technique. The superior hypogastric plexus and nerves are only involved during the abdominal portion of taTME and are of no concern during transanal dissection. The pelvic splanchnic nerves are composed of the anterior rami of sacral nerves 2−5 and provide parasympathetic innervation to the hindgut. Their initial course lies deep to the parietal fascia, however, as they ascend to join the inferior hypogastric plexus, and then they pierce the endopelvic fascia and ultimately cross the retrorectal space to form branches into the rectum through the lateral ligaments. During taTME, the lower splanchnic nerves are commonly susceptible to injury during the initial dissection and identification of the appropriate mesorectal plane (Figure 7). Following full thickness proctotomy, clear recognition of the endopelvic fascia at the 4- and 8-o’clock position avoids early lateral exposure of the splanchnic nerves. Visualization of levator ani muscle fibers is the earliest clue that dissection has proceeded deep to the endopelvic fascia, placing the nerves at risk. Further mobilization cephalad requires dissection just on the fascia propria of the rectum to avoid injury to the nerves as they lay on top of the endopelvic fascia and are often tented in by the retracted rectum.
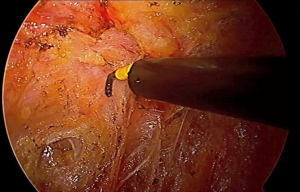
The inferior hypogastric plexus in the lower third of the rectum is formed from a coalescence of distal afferent fibers from the sympathetic hypogastric trunks as they descend, and pelvic parasympathetic fibers from the splanchnic nerves. Fibers from the inferior hypogastric plexus innervate the seminal vesicles, prostate, bladder, cervix and vagina. These nerves are responsible for penile erection, detrusor contractility, female arousal, and vaginal lubrication. Dissection of the extraperitoneal rectum too laterally during conventional TME has been well described as a cause of plexus injury and sexual dysfunction. With taTME, excessive lateral dissection is a common occurrence, especially early in the learning curve, exposing the plexus to increased injury. Dissection often can proceed unnoticed well up the sidewall of the obturator internus, lateral to the pelvic plexus and obturator nodal packet. During corrective maneuvers to adjoin with the anterior plane, the entire neurovascular bundle is at risk for injury. During this improper maneuver, dissection is carried laterally to the S3 nerve trunk which can be inadvertently transected with corrective actions. Dissection anterior to Denonvilliers’ fascia, classically described by Heald, is also speculated to increase risk of nerve injury and has evolved to selectively dissect posterior to the fascia based on tumor location and concern over circumferential resection margins. Similarly, during taTME, the surgeon can choose the appropriate level of dissection, taking special caution anterolaterally where the neurovascular bundles (of Walsh) are more anatomically accessible. Thus, correct total mesorectal dissection and appreciation of the course of the pelvic nerves is paramount for nerve preservation.
Anastomotic leak
Anastomotic leak following low pelvic anastomosis leads to sepsis, decreased survival, inability to administer adjuvant chemotherapy, poor function, and often permanent stoma. Despite the feelings by many taTME surgeons that a double pursestring anastomosis could be superior, a review of the international taTME registry recently suggested increased rates of anastomotic leak following taTME (7). Although this certainly reflects the learning curve and early adoption of taTME, two principal factors are unique to taTME and account for the change in technique required for successful anastomosis. Unlike conventional surgery where the distal rectum is transected with an endoscopic linear stapler, at the completion of taTME an open distal anorectal cuff remains. The anorectal cuff should be secured with a pursestring as tightly as possible. Secondly, a central opening always remains, even after a secure closure, as it is virtually impossible to close the stump completely. A fibrotic or immobile cuff may be difficult to close. Dissenting opinion exists over whether the distal cuff should be mobilized further either manually or endoscopically. Full thickness suturing of the distal rectal cuff will help prevent from inadvertently tearing through during pursestring tightening.
More importantly, it is mandatory the anvil of the stapler comes directly through the central defect. Alternatively, hand-sewn techniques can be utilized for a fraction of the cost. Multiple different transanal anastomotic techniques and their pitfalls have been described and should be reviewed by taTME surgeons. The transanal anastomosis requires several steps, unique from the conventional approach, and should be studied and practiced.
Urethral injury
Of all the possible complications, urethral injury is a main deterrent to forego attempts at a taTME and is certainly the most unique of this procedure (8). Permanent urinary diversion is a common sequalae, as healing in the radiated field is challenging, especially with complete transection (9). Furthermore, access to repair is only possible from a transanal approach. Visual identification of the ureters, whether during an open or laparoscopic transabdominal approach for low pelvic dissections, has decreased the rate of injury significantly. The use of stents to identify the ureters can further protect from injury. During taTME, the ureters are not exposed during dissection of the extraperitoneal rectum. However, both the bladder and urethra have been injured with increasing incidence. Intraoperative markers of erroneous dissection planes, such as bleeding or visualization of striated muscle, must be recognized and corrected. Advanced energy devices are not recommended as they create the possibility to seal larger vessels and structures with impunity not generally encountered in a proper dissection. The correct dissection plane will require minimal electrocautery, as loose areolar tissue should easily be bluntly dissected.
The urethra lies anterior to the prostate, which encompasses it from behind. Therefore, injury to the urethra absolutely requires improper dissection anterior to the prostate, first dropping the prostate down, and subsequently, forcing the distal membranous urethra to assume a vertical orientation. For this reason, the authors have adopted an anterior first approach, allowing early identification and entry into the rectoprostatic plane (Figure 8A,B,C). In addition, dissecting posteriorly first, and then following the plane laterally to finish anteriorly can lead to prostatic mobilization by initial excessive lateral dissection. Be wary of encountering any large vascular structures, as the anterior mesorectum is usually quite thin, even in obese patients, and rarely has large vessels within it. Observation of this should prompt immediate caution. In the same vein, the presence of a bulky midline structure should be immediately alarming and prompt immediate reevaluation.
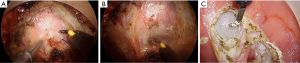
Urethral injury is much more likely to result from intersphincteric dissection of distal rectal tumors, as plane identification, and dissection deep and too lateral to the endopelvic fascia is more common. Radiation induced fibrosis and edema, as well as anterior tumors increase difficulty. At this level, the mesorectum is absent and requires dissection directly on the anorectal tube. Dissection too anterior leads to early mobilization of the prostate, thus dropping it down. The use of illuminated ureteral catheters, indocyanine green, and frequent palpation or manipulation of the urinary catheter have all been utilized to maintain orientation and avoid urethral injury (10-12).
Air embolism
Carbon dioxide or gas embolism is a life-threatening complication with low incidence in laparoscopic surgery. Its occurrence in laparoscopic and robotic total mesorectal excision has previously not been described. Intraoperatively it is marked by an acute drop in end tidal CO2, O2 saturation, followed by hypotension and cardiovascular collapse. Several recent case reports of taTME associated with gas embolism have been described, including the most recent publication demonstrating 25 cases collaboratively amongst the international taTME working group (13). Continuous high flow insufflation at pressures >15 mmHg were noted in 24 of 25 operations. However, in the registry in which the data was obtained, few of the operations utilized alternative insufflators. It has been recommended that insufflation pressures be set at 12 mm or less. Additionally, more than 50% of the cases were associated with a tangential injury to an anterior pelvic vessel, such as a vaginal or periprostatic vein. Early recognition is critical to treatment and resuscitative efforts. Gas bubbles can occasionally be directly seen in conjunction with blood escaping from a venous injury (Figure 9). Anesthesiologists treating patients undergoing taTME should be made well aware of the potential occurrence of this complication. Acute drop in end tidal CO2 is easily recognized by the anesthetist and is nearly pathognomonic for gas embolism, prompting immediate treatment. Insufflation by the operating team should be immediately ceased in conjunction with wound packing, and/or filling the operative field with saline. Transesophageal echocardiography availability is justified for early diagnosis as well, but with aggressive support measures, cardiovascular status is often restored quickly, and the operation can usually commence. taTME utilized for reoperative pelvic surgery, an increasing indication for taTME, further complicates transanal anatomy leading to higher risks of wrong plane surgery and subsequent venous injury.

Acknowledgement and education of this important risk allows adequate preparation by the surgical team for prevention, early detection, and treatment. The use of a transanal port with a gas-tight seal, and operating at high pressures in a small volume cavity, pose an inherent risk of taTME. High flow insufflators can be utilized, but with lower insufflation pressures. Alternative insufflators are sufficient in conjunction with an insufflation stabilization bag (ISB) (Applied Medical, Santa Margarita). Prevention is the best solution, with quick control of any bleeding, and consideration for lowering the CO2 insufflation when there is any hemodynamic concern. Avoidance of transanal dissection in the incorrect anatomic plane with subsequent venous injury is critical to grasp prior to undertaking this operation.
Conclusions
TaTME represents one of the latest advancements in minimally invasive colorectal surgery that has the potential for both improved oncologic and functional results as well as crippling complications. TaTME is a challenging procedure, combining advanced laparoscopic skills, new planes, and unfamiliar perspectives. Critical understanding of pelvic anatomy and the effects of pneumopelvis is required. Case observation, cadaveric practice, as well as systematic learning and teaching, is needed for widespread success. We have reviewed critical possible traps in the technique and provided methods to avoid these. These important technical pearls will aid the novice and intermediate surgeon in reducing the morbidity associated with the learning curve of this admittedly difficult technique.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Satish Kumar Warrier and Alexander Heriot) for the series “taTME” published in Annals of Laparoscopic and Endoscopic Surgery. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ales.2020.01.02). The series “taTME” was commissioned by the editorial office without any funding or sponsorship. MA reports personal fees, non-financial support and other from Applied Medical, personal fees and non-financial support from Stryker Endoscopy, personal fees from Conmed, personal fees and other from Human Extensions, personal fees from Cooper Surgical, outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Muratore A, Mellano A, Marsanic P, et al. Transanal total mesorectal excision (taTME) for cancer located in the lower rectum: short- and mid-term results. Eur J Surg Oncol 2015;41:478-83. [Crossref] [PubMed]
- Penna M, Hompes R, Arnold S, et al. Transanal Total Mesorectal Excision: International Registry Results of the First 720 Cases. Ann Surg 2017;266:111-7. [Crossref] [PubMed]
- Ranbarger KR, Johnston WD, Chang JC. Prognostic significance of surgical perforation of the rectum during abdominoperineal resection for rectal carcinoma. Am J Surg 1982;143:186-8. [Crossref] [PubMed]
- Ohtani H, Tamamori Y, Azuma T, et al. A meta-analysis of the short-and long-term results of randomized controlled trials that compared laparoscopy-assisted and conventional open surgery for rectal cancer. J Gastrointest Surg 2011;15:1375-85. [Crossref] [PubMed]
- Xiong B, Ma L, Zhang C, et al. Robotic versus laparoscopic total mesorectal excision for rectal cancer: a meta-analysis. J Surg Res 2014;188:404-14. [Crossref] [PubMed]
- Denost Q, Adam JP, Rullier A, et al. Perineal transanal approach: a new standard for laparoscopic sphincter-saving resection in low rectal cancer, a randomized trial. Ann Surg 2014;260:993-9. [Crossref] [PubMed]
- Penna M, Hompes R, Arnold S, et al. Incidence and risk factors for anastomotic failure in 1594 patients treated by transanal total mesorectal excision: results from the international TaTME registry. Ann Surg 2019;269:700-11. [Crossref] [PubMed]
- Bjørn MX, Perdawood SK. Transanal total mesorectal excision--a systematic review. Dan Med J 2015;62:A5105 [PubMed]
- Sylla P, Knol JJ, D'Andrea AP, et al. Urethral Injury and Other Urologic Injuries During Transanal Total Mesorectal Excision: An International Collaborative Study. Ann Surg 2019; [Epub ahead of print]. [Crossref] [PubMed]
- Atallah S, Martin-Perez B, Albert M, et al. Transanal minimally invasive surgery for total mesorectal excision (TAMIS-TME): results and experience with the first 20 patients undergoing curative-intent rectal cancer surgery at a single institution. Tech Coloproctol 2014;18:473-80. [Crossref] [PubMed]
- Atallah S, Martin-Perez B, Drake J, et al. The use of a lighted stent as a method for identifying the urethra in male patients undergoing transanal total mesorectal excision: a video demonstration. Tech Coloproctol 2015;19:375. [Crossref] [PubMed]
- Barnes TG, Volpi D, Cunningham C, et al. Improved urethral fluorescence during low rectal surgery: a new dye and a new method. Tech Coloproctol 2018;22:115-9. [Crossref] [PubMed]
- Dickson EA, Penna M, Cunningham C, et al. Carbon Dioxide Embolism Associated With Total Mesorectal Excision Surgery: A Report From the International Registries. Dis Colon Rectum 2019;62:794-801. [Crossref] [PubMed]
Cite this article as: Albert M, Aka AA. Complication profile of transanal total mesorectal excision and how it differs. Ann Laparosc Endosc Surg 2020;5:28.


