
Energy in robotic surgery
Introduction
Robotic surgery has become a mainstay in operating rooms throughout the United States and the world. Since its initial approval by the US Food and Drug Administration (FDA) for urologic procedures in 2000, the use of robotics has grown exponentially across different surgical fields and has become the standard of care for prostatectomy (1-5). Although other robotic platforms are finding their way into operating rooms across the nation, the da Vinci platform (Intuitive Surgical, Sunnyvale, CA) is the most common platform in use (6,7). This technologic advancement could not be maintained without the development and use of specialized energy instruments and electrosurgical generators which are used to cut tissue and achieve hemostasis. The devices used during robotic surgery are similar to those used during minimally invasive and open surgery however there are several important differences. This review provides an overview of the currently available energy devices and their optimal use in robotic surgery. We also discuss the impact of the robotic platform on the eight patterns of injury from energy devices. Of note, as robotic surgery is currently dominated by the da Vinci Si and Xi platforms, the majority of our review is focused on the associated, approved devices and their fit and function within those platforms.
Electrosurgical generators
Radiofrequency energy for tissue dissection and hemostasis is delivered by an electrosurgical generator. There are many generators available for use with the S/Si systems (Table 1, Figure 1). While other electrosurgical units (ESUs) could be used, there are limited cables and adapters that are approved for use with the proprietary instruments and accessories. The choice of a specific ESU can be further exacerbated by the use of the robotic platform by multiple surgical subspecialties with different preferences for energy devices.
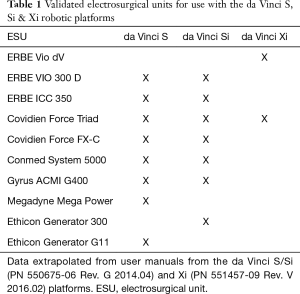
Full table

The choice of generator was significantly narrowed with the Xi system as it included an integrated generator produced in conjunction with ERBE—ERBE VIO dV (ERBE USA, Marietta, GA; Intuitive, Sunnyvale, CA). However, in order to maintain compatibility with the most common alternative, the Xi is also certified for use with the Covidien (formerly Valleylab) ForceTriad (Covidien-Medtronic, Boulder, CO). Both of these generators provide monopolar, bipolar and advanced bipolar energy options. Intuitive received FDA approval in late November 2019 for a new proprietary generator, the “E-100,” for use with the da Vinci advanced sealing devices (Vessel Sealer Extend® and the unreleased SynchroSeal® instrument). Based upon the 510(k) application this ESU does not provided monopolar functions and is designed for use alongside either the integrated ERBE VIO dV or the Covidien ForceTriad with the Xi platform.
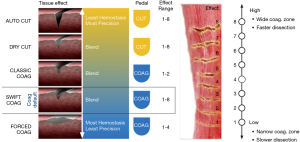
The VIO dV and ForceTriad are distinctly different ESUs and the choice between them for current practitioners is made difficult by competing specialty preferences, hospital contracting and the overall equivalent safety profiles. The integrated VIO dV is unfamiliar to many surgeons as its most similar counterpart, the VIO 300d, is rarely found outside of gastroenterology suites in the United States. In contrast, the ForceTriad enjoys significant market presence in the operating room and its predecessor, the Force FX, remains a common site in the US. The energy settings and modalities are similar in that there are two overarching waveforms; one designed to cut or transect tissue (“cut” modes) and one designed to achieve more hemostasis (while still able to transect tissue), “coag” mode.
Significant differences arise thereafter as the VIO dV includes additional waveforms such as “swift coag,” “forced coag,” etc. that provided blended waveforms which respond differently to tissue resistance (impedance) as they are being used. Beyond the energy modality choice, the VIO dV requires the selection of a “tissue effect” as well as a “power limit.” The myriad of possible tissue effects are illustrated in the quick reference guide available online (Figure 2). In contrast, the ForceTriad utilizes a simpler platform offering the selection of power (W) and 4 modes: “pure,” or “blend” cut modes and fulgurate, “fulg,” or “spray” coag modes. In brief, the blended cut mode adds hemostasis to pure cut; the “spray” coag mode (vs. “fulg”) is intended for use on large areas where superficial hemostasis is required (e.g., liver). Approximate power equivalencies are listed in Table 2.
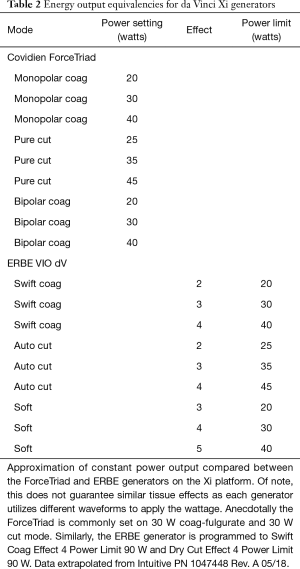
Full table
Due to the different energy waveforms, a simple translation of power settings does not necessarily result in similar tissue effect. Anecdotally, the most common monopolar energy settings when using the ForceTriad are 30 W fulg coag mode and 30 W pure cut mode. In our experience, the most common ERBE settings are swift coag effect 4 power limit 90 W and dry cut effect 4 power limit 90 W. Additional fine tuning is available within each generator and should be guided by the desired tissue effect (for example, if more hemostasis needed, increase the wattage to 40 or tissue effect to 5). Surgeon preferences can also be saved in the VIO dV.
With respect to safety, both are FDA approved and have similar safety profiles yet remain susceptible to stray energy injuries (as do all ESUs) (8). For example, recent testing in endoscopy suggest that the constant voltage regulation mechanism of the ERBE VIO 300 results in less stray energy transfer (9,10). Study by our group in the Si platform demonstrated similar findings during open air activation (i.e., activating the device without touching tissue) (Table 3). Techniques to reduce the risk of stray energy transfer and discussed in detail below.

Full table
Energy application: monopolar
Devices
Monopolar energy application via a hook is one of the most common instruments used by laparoscopic and robotic surgeons. The hook allows the surgeon to dissect and precisely apply energy to adhesions, small blood vessels and lymphatics. The “heel” of the device can also be used to apply energy in a sweeping motion allowing division and hemostasis of tissue in a large area more efficiently. This versatile instrument is central to the safe and hemostatic performance of minimally invasive cholecystectomy (11).
The robotic equivalent utilizes EndoWrist® (Intuitive) technology (Figure 3A). This is utilized in most robotic instruments and allows manipulation in 570°. This exceeds the human wrist and facilitates fine dissection in restricted spaces and precise planes. The hook is primarily used in robotic cholecystectomy and inguinal hernia repair (Figure 3B). The second most commonly used monopolar instrument in laparoscopic and robotic surgery is the scissors (Figure 4A,B). This device also utilizes EndoWrist® technology again allowing for precise dissection, especially in restricted spaces.
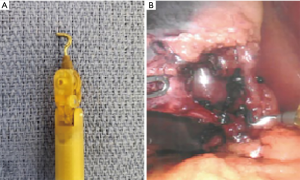
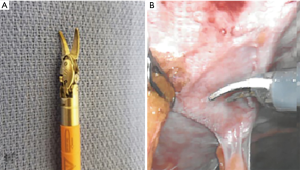
Both of these energy devices are made more effective with the magnification and 3-Dimensional visualization provided by the da Vinci 3DHD Camera Endoscope. The increased articulation and precision have made the energized scissors extremely effective for dissection and hemostasis. These are commonly used for lysis of adhesions and more complex dissection such as in the pelvis during low anterior and abdomino-pelvic resections as well as inguinal and ventral hernia repairs (Figure 5) (12).

The last monopolar instrument available for the robotic platform is a spatula (Figure 5). This includes EndoWrist® technology and provides efficient, superficial tissue desiccation over a wide area. This, for example, is used during liver resections to achieve hemostasis of the superficial vessels after tissue division (13).
Complications
Energy use in the operating room is associated with 8 reproducible patterns of electrosurgical injury: fires, antenna coupling, insulation failure, residual heat, direct application, interaction with electronic devices, direct coupling and capacitive coupling (14). Data is lacking with regard to the impact of the robotic platform on these patterns of injury however inferences can be made to laparoscopy. The incidence of surgical fires should be no different than traditional laparoscopy and will not be reviewed further. Direct coupling and direct application injuries are more common in open surgery and will also not be discussed. No data is available with respect to interference with electronic devices. Antenna coupling, capacitive coupling, insulation failure and residual heat will be discussed where applicable below.
The transfer of stray energy via antenna and capacitive coupling is of particular interest as both of these injury patterns are made worse by large diameter metallic objects that are in close proximity to the energy device (9,15-19). This is unavoidable and theoretically increases the risk of stray energy injuries in robotics. Ex vivo data from our lab demonstrate comparable stray energy transfer to laparoscopy during open air activation with the ForceTriad. This is significantly reduced by reducing the power setting from 30 to 15 W in coag mode or utilizing a low voltage mode such as pure or blended cut (Table 4). In addition, the ERBE ESU created significantly less stray energy transfer at baseline as well; the exact mechanism of which is likely related to constant voltage regulation in contrast to the constant power regulation of the ForceTriad. More study is needed to confirm these findings and delineate their cause.

Full table
Reducing the power settings and/or utilizing low voltage modes are effective techniques to reduce stray energy transfer but special emphasis must remain on the avoidance of open air activation (e.g., stepping on the pedal before the hook or scissors is touching tissue). In our lab, no significant energy was transferred when the monopolar device was activated while contacting tissue (Table 3). This finding is logically sound as, once the energy device is activated, radiofrequency energy is delivered to the metal L-hook, scissor or spatula and seeks to complete the circuit (return to the dispersive electrode or “grounding pad”). If no circuit can be found, then the energy “builds up” until it can jump across insulation (capacitive coupling) or transfer into nearby metal objects (antenna coupling).
Another important mechanism of energy complication is insulation failure. Vascular and bowel injuries have occurred as a result of stray energy injury and none are more obvious than when they occur via an insulation defect (20,21). The increased angulation and versatility of the EndoWrist® is facilitated by a rubber tip cover accessory when the scissor is used. A study by Espada et al. found that nearly 80% of robotic instruments developed an insulation failure after 10 uses (22). In theory, the increased angulation and versatility of the cover tip accessory results in micro tears that can develop into insulation defects. This was detected in up to 39% of cases after a single use with the first-generation tip (23). A second generation cover demonstrated no insulation defects after first time use however no additional studies have been published. As the surgeon, visual inspection remains the most effective way to evaluate the tip cover accessory and should be done prior to use in every case.
In sum, there remains minimal data to guide clinicians in the avoidance of monopolar energy-based complications during robotic surgery. Extrapolation from laparoscopy reveals the following recommendations: (I) utilize the lowest power setting (watts) to achieve the desired tissue effect. (II) Utilize low voltage modes (ERBE swift coag vs. forced coag; ForceTriad blend mode vs. coag mode) whenever possible. (III) Avoid open air activation. (IV) Inspect the insulation prior to use.
Energy application: bipolar
The robotic platform has an array of bipolar instruments which can serve as both dissectors and tissue graspers as well as achieve hemostasis and transect tissue. The most commonly used is the standard bipolar grasper which offers a Maryland or Fenestrated tip (Figure 6A,B). This also utilizes the EndoWrist® technology and has become a versatile instrument for grasping and retracting tissue that doubles as a coating device to achieve hemostasis of small vessels.
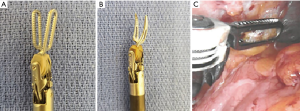
The bipolar forceps (Figure 6C) are particularly useful when used alongside a scissor for hemostasis of small vessels that were incompletely controlled during scissor dissection. Settings for the ForceTriad are typically similar to monopolar (30 W coag mode) as bipolar itself cannot cut tissue (this requires an integrated blade such as the Vessel Sealer® discussed below). The integrated VIO dV bipolar modes are adjustable similar to the monopolar modes with tissue effect (1-8) and an adjustable power limit. While a “cut” mode is offered, this is rarely used. A built-in safety mechanism, “Autostop,” ensures that the bipolar energy can only be activated with an adequate amount of tissue is grasped (as measured by tissue impedance). This can limit the device’s effectiveness for very small vessels.
An advanced bipolar device is available for the sealing of vessels up to 7 mm in diameter, the Vessel Sealer ® and the updated version, the Vessel Sealer Extend®. Similar to Ligasure (Covidien-Medtronic, Boulder, CO) the Vessel Sealer combines advanced bipolar vessel sealing with the versatility of EndoWrist® (Figure 7). This allows dissection and advanced vessel sealing, which can be of particular use within small spaces such as the pelvis or mediastinum, without the need to change instrument, thus reducing operative time (24). Of note, however, Kong et al. encountered tip inactivation when using the Vessel Sealer® for both dissection and hemostasis requiring instrument exchange for the standard bipolar device (25).
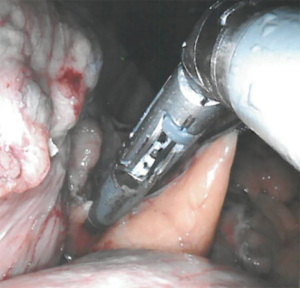
The Vessel Sealer Extend® increases the versatility of the original device with 60° of fully wristed articulation and the ability to seal vessels up to 7 mm in size at a 90° angle. This is offered in a 30% slimmer device that includes 3 mm of additional sealing space. The authors have not yet trialed this device however it does appear to be a significant improvement from the original version.
Energy application: ultrasonic
An ultrasonic device is available for use in the robotic platform through a partnership with Ethicon (ACE Harmonic). This provides vessel sealing and transection up to 7 mm in size (26,27). However, due to the device construction requiring conversion of radiofrequency energy into an ultrasonic, vibrating tip, it lacks the flexibility of EndoWrist®. Thus it has not been significantly adopted despite the decreased thermal spread and reduced coagulum and charring that is found with ultrasonic devices.
Other robotic platforms
While our focus has been on the most widely available robotic platforms (da Vinci S/Si and Xi), there are multiple other companies that are in the final stages of development of surgical robots. The only FDA approved device that has seen limited release in the US is the Senhance® Surgical System by Transenterix. It brings new features to the field of robotic surgery such as eye tracking capability and haptic feedback. Given the limited release, no significant statements can be made regarding the associated energy devices.
Conclusions
Robotics has become an essential part of surgical science and its use is rapidly expanding. Surgeons must have a clear understanding of the instruments and accessories available in order to operate with optimal effect and efficiency. This requires an understanding of the use of radiofrequency energy to achieve tissue dissection, transection and hemostasis. It is our hope that this brief review provides the groundwork to understand the different monopolar, bipolar and ultrasonic energy options as well as the risks, benefits and alternatives to each. While more study is needed, adherence to the basic guidelines outlined above will result in improved efficiency as well as reduce patient injury.
Acknowledgments
The authors would like to thank Drs. Alyssa Peace and Paul Montero for helping with preparation of photographs.
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ales.2020.03.06). Dr. KJW reports non-financial support from Intuitive Surgical, outside the submitted work. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work and in ensuring that questions related to the accuracy or integrity of any part of the work presented have been appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kelly S. Intuitive Surgical profit up on strong da Vinci robot sales: MeditechDive; 2018. Available online: https://www.medtechdive.com/news/intuitive-surgical-profit-up-on-strong-da-vinci-robot-sales/528257/
- Tan A, Ashrafian H, Scott AJ, et al. Robotic surgery: disruptive innovation or unfulfilled promise? A systematic review and meta-analysis of the first 30 years. Surg Endosc 2016;30:4330-52. [Crossref] [PubMed]
- Leal Ghezzi T, Campos Corleta O. 30 Years of Robotic Surgery. World J Surg 2016;40:2550-7. [Crossref] [PubMed]
- Estey EP. Robotic prostatectomy: The new standard of care or a marketing success? Can Urol Assoc J 2009;3:488-90. [Crossref] [PubMed]
- Laviana AA, Williams SB, King ED, et al. Robot assisted radical prostatectomy: the new standard? Minerva Urol Nefrol 2015;67:47-53. [PubMed]
- Peters BS, Armijo PR, Krause C, et al. Review of emerging surgical robotic technology. Surg Endosc 2018;32:1636-55. [Crossref] [PubMed]
- Gosrisirikul C, Don Chang K, Raheem AA, et al. New era of robotic surgical systems. Asian J Endosc Surg 2018;11:291-9. [Crossref] [PubMed]
- Karacan T, Usta T, Ozkaynak A, et al. Comparison of the Thermal Spread of Three Different Electrosurgical Generators on Rat Uterus: A Preliminary Experimental Study. Gynecol Obstet Invest 2018;83:388-96. [Crossref] [PubMed]
- Jones EL, Madani A, Overbey DM, et al. Stray energy transfer during endoscopy. Surg Endosc 2017;31:3946-51. [Crossref] [PubMed]
- Mendez-Probst CE, Vilos G, Fuller A, et al. Stray electrical currents in laparoscopic instruments used in da Vinci(R) robot-assisted surgery: an in vitro study. J Endourol 2011;25:1513-7. [Crossref] [PubMed]
- Pontarelli EM, Grinberg GG, Isaacs RS, et al. Regional cost analysis for laparoscopic cholecystectomy. Surg Endosc 2019;33:2339-44. [Crossref] [PubMed]
- Cui R, Yu MH, Chen JJ, et al. Monopolar Electrosurgical Scissors Versus Harmonic Scalpel in Robotic Anterior Resection of Rectal Cancer: A Retrospective Cohort Study. J Laparoendosc Adv Surg Tech A 2019;29:880-5. [Crossref] [PubMed]
- Holloway RW, Brudie LA, Rakowski JA, et al. Robotic-assisted resection of liver and diaphragm recurrent ovarian carcinoma: description of technique. Gynecol Oncol 2011;120:419-22. [Crossref] [PubMed]
- Jones EL, Dean J. Surgical Energy. In: Fischer JE, Ellison EC, Upchurch GR, et al. editor. Fischer's Mastery of Surgery. 1. 7 ed. Philadelphia: Wolters Kluwer; 2018.
- Jones EL, Robinson TN, McHenry JR, et al. Radiofrequency energy antenna coupling to common laparoscopic instruments: practical implications. Surg Endosc 2012;26:3053-7. [Crossref] [PubMed]
- Robinson TN, Jones EL, Dunn CL, et al. Separating the Laparoscopic Camera Cord From the Monopolar "Bovie" Cord Reduces Unintended Thermal Injury From Antenna Coupling: A Randomized Controlled Trial. Ann Surg 2015;261:1056-60. [Crossref] [PubMed]
- Overbey DM, Townsend NT, Chapman BC, et al. Surgical Energy-Based Device Injuries and Fatalities Reported to the Food and Drug Administration. J Am Coll Surg 2015;221:197-205.e1. [Crossref] [PubMed]
- Overbey DM, Hilton SA, Chapman BC, et al. Hand-to-hand coupling and strategies to minimize unintentional energy transfer during laparoscopic surgery. J Surg Res 2017;219:103-7. [Crossref] [PubMed]
- Townsend NT, Jones EL, Paniccia A, et al. Antenna coupling explains unintended thermal injury caused by common operating room monitoring devices. Surg Laparosc Endosc Percutan Tech 2015;25:111-3. [Crossref] [PubMed]
- Cormier B, Nezhat F, Sternchos J, et al. Electrocautery-associated vascular injury during robotic-assisted surgery. Obstet Gynecol 2012;120:491-3. [Crossref] [PubMed]
- Martin KE, Moore CM, Tucker R, et al. Quantifying inadvertent thermal bowel injury from the monopolar instrument. Surg Endosc 2016;30:4776-84. [Crossref] [PubMed]
- Espada M, Munoz R, Noble BN, et al. Insulation failure in robotic and laparoscopic instrumentation: a prospective evaluation. Am J Obstet Gynecol 2011;205:121.e1-5. [Crossref] [PubMed]
- Engebretsen SR, Huang GO, Wallner CL, et al. A prospective analysis of robotic tip cover accessory failure. J Endourol 2013;27:914-7. [Crossref] [PubMed]
- Ortenzi M, Ghiselli R, Baldarelli M, et al. Is the bipolar vessel sealer device an effective tool in robotic surgery? A retrospective analysis of our experience and a meta-analysis of the literature about different robotic procedures by investigating operative data and post-operative course. Minim Invasive Ther Allied Technol 2018;27:113-8. [Crossref] [PubMed]
- Kong SH, Kim TH, Huh YJ, et al. A Feasibility Study and Technical Tips for the Use of an Articulating Bipolar Vessel Sealer in da Vinci Robot-Assisted Gastrectomy. J Laparoendosc Adv Surg Tech A 2017;27:1172-9. [Crossref] [PubMed]
- Amaral JF. The experimental development of an ultrasonically activated scalpel for laparoscopic use. Surg Laparosc Endosc 1994;4:92-9. [PubMed]
- Aykan Yuksel B, Karadag B, Mulayim B. Comparison of the efficacy and safety of two advanced vessel sealing technologies in total laparoscopic hysterectomy. J Obstet Gynaecol Res 2019;45:2220-7. [Crossref] [PubMed]
Cite this article as: Wikiel KJ, Robinson TN, Jones EL. Energy in robotic surgery. Ann Laparosc Endosc Surg 2021;6:9.


