The robotic approach for vascular and endovascular procedures: a narrative review
Introduction
Over the last several years, robot technology has greatly expanded the field of general surgery. As the technology has evolved, providers have continued to search for and experiment with new applications within a myriad of surgical specialties and procedures. This has led to new and innovative applications for robotic technology in both the field of vascular surgery as well as in procedures requiring vascular expertise. The sentinel example of a minimally invasive technique within the field of vascular surgery occurred in 1993, when Dion et al. performed the first laparoscopic-assisted aortobifemoral bypass (1,2). While the first use of a minimally invasive technique was cited in 1993, there is very little substantive knowledge in today’s world detailing how robot technology may be utilized within the field of vascular surgery. The goal of this brief narrative review is to provide an overarching view of the advances made in robotic-assisted interventions within the field of vascular surgery. This review will be largely split into two parts. Part I will broadly explore robot technology as it pertains to classically open vascular surgery procedures. Part II will overview how robot technology has further advanced endovascular interventions. This review is not designed to provide details on exact robotic approaches, nor how those approaches behaved; rather, this is a broad narrative review on utilization of robot technology within the field of vascular surgery. We are proud to present this article in accordance with the Narrative Review reporting checklist (available at https://ales.amegroups.com/article/view/10.21037/ales-23-24/rc).
Methods
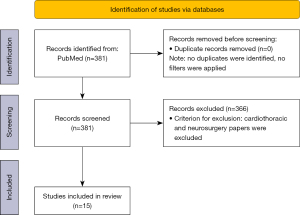
A comprehensive literature search was employed using the following search strategy on PubMed: (“Robotic Surgical Procedures”[Mesh]) AND (“Vascular Surgical Procedures”[Mesh]). The formula we used is shown in Table 1. We excluded articles involving cardiac and neurosurgery. The reason for those exclusions is that the authors were most interested in performing this literature review from the focus of the vascular surgeon, and procedures involving the heart and brain are outside his or her scope of practice. All articles were then screened manually, and then the final selection was determined by the primary authors. The articles were specifically selected on the basis of their ability to provide a synopsis of the advances made in robotic-assisted interventions within the field of vascular surgery. More details of the method are shown in Table 1 and Figure 1.
Table 1
| Items | Specification |
|---|---|
| Date of search | 01/01/2023 to 03/01/2023 |
| Databases and other sources searched | PubMed |
| Search terms used | “Robotic Surgical Procedures”[Mesh] AND “Vascular Surgical Procedures”[Mesh] |
| Timeframe | 2002–2022 |
| Inclusion and exclusion criteria | Inclusion criteria: research articles and reviews in English about vascular surgery and robotics. Exclusion criteria: articles related to cardiothoracic and neurosurgery |
| Selection process | AHV conducted the search. All authors then voted to obtain consensus on which articles would be included |
| Any additional considerations, if applicable | Some papers were identified by reviewing reference lists of relevant publications |
Part I: open procedures
Aortoiliac reconstruction
Since the introduction of endovascular therapies, the field of vascular surgery has grown tremendously over the last 100 years. While an endovascular approach has fundamentally altered the field of vascular surgery, one principle holds true—at least for now—that there remains a select cohort of patients who require open vascular reconstruction. While various principles exist to guide the use of an open versus an endovascular strategy, there needs to remain a patient-centered approach in reconstruction planning. One such classification system that helps to guide reconstruction planning is the TransAtlantic Inter-Society Consensus (TASC) guidelines. According to the most recent TASC guidelines (published in 2007), endovascular surgery is the treatment of choice for TASC A, B and some C lesions, but with diffuse TASC D aortoiliac lesions, the gold standard remains open grafting (3). Management of TASC D lesions is traditionally performed in an open fashion, but in 2002, Wisselink et al. performed the first robot-assisted aortobifemoral bypass (4). This procedure was technically successful and resulted in a good outcome thereby showing the utility of incorporating robot technology into vascular reconstruction procedures.
In the years since, several case series have been published evaluating robot-assisted aortoiliac procedures. The largest of these series was a single-center retrospective review in 2016 by Štádler et al. evaluating 298 of 310 patients who successfully underwent a variety of robot-assisted aortic procedures in Prague, Czech Republic (5). Sixty-one patients underwent abdominal aortic aneurysm (AAA) repair, 224 had aortoiliac reconstruction and the remaining 24 had other procedures, such as splenic artery aneurysm repair, type II endoleak repair, and median arcuate ligament release. Inclusion criteria were AAA measuring at least 5.5 cm, iliac artery aneurysm measuring at least 3 cm and either TASC C or D lesions. The majority of cases, 96.8% (300/310), were able to be completed by maintaining a robotic approach, while the 10 remaining cases required conversion to laparotomy. The most common complication cited for conversion was bleeding at the site of the anastomosis. The mortality rate was 0% in the aortic occlusive disease subgroup, and 1.6% in the aortoiliac aneurysm subgroup. Some of the other relevant factors that this case series reported on were median operative time of 204 minutes (range, 150–360 minutes), clamp time of 49 minutes (range, 21–120 minutes), estimated blood loss of 571 mL (range, 50–3,500 mL), ICU stay of 1.2 days (range, 1–5 days), and hospital stay of five days (range, 4–10 days).
Overall, when compared to the studies published by Coggia et al. (6), this case series highlights that robot-assisted AAA repair, as compared to laparoscopic repair, has an improved median operating time (204 versus 290 minutes), clamp time (49 versus 78 minutes), estimated blood loss (571 versus 1,680 mL) and mortality rate (0% versus 6.1%) (4). This case report highlights that a minimally invasive robot-assisted approach is a feasible, safe and effective option for vascular reconstruction.
First rib resection
While the robot-assisted approach has been primarily studied in aortoiliac reconstruction, there are a number of other applications as well. In 2019, Gharagozloo et al. conducted a retrospective single-center study review of 83 patients with Paget-Schroetter syndrome (PSS) (7). The treatment algorithm for PSS generally involves anticoagulation followed by decompression of the thoracic outlet, most commonly via an open first rib resection. The patients in this case series were treated in the standard fashion with first thrombolysis, but then underwent transthoracic robot-assisted resection of the medial aspect of the first rib. The study reported operative times of 127±20.8 minutes with no intraoperative complications or injury to the subclavian vessels during dissection, no neurovascular complications, and no mortality at 90 days. This minimally invasive approach suggests that robotic first rib resection is a promising application of the minimally invasive approach, as it avoids the neurogenic complications which are widely reported following open resection (7).
Venous thrombectomy
Another common vascular procedure, wherein robot technology has proven to be successful, is venous thrombectomy—especially when the clot extends into the inferior vena cava (IVC). For example, Wang et al. published a case report of 13 patients who underwent robot-assisted IVC thrombectomy in Beijing, China. These 13 patients developed tumor thrombi that extended above the hepatic veins (8). Treatment for these thrombi typically involves liver mobilization and clamping of the porta hepatis with both suprahepatic and infradiaphragmatic exposure (9). In this case report, Wang et al. demonstrated the utility of robot assistance and reported a median operative time of 465 minutes (range, 338–567 minutes), blood loss of 2,000 mL (1,000–3,000 mL) and a perioperative mortality rate of 7.7%. At the 18-month follow up, there were two additional deaths noted. Overall, this case report demonstrates how a minimally invasive approach may be utilized in treating complex vascular disorders that are not easily amenable to an open approach (8).
Venous reconstruction in complex oncologic resection
There have also been case reports of surgeons utilizing the robot to successfully reconstruct the portal vein in complex oncologic resections, most commonly during total gastrectomy and pancreaticoduodenectomy (10,11). In 2019, Beane et al. published a retrospective review of 50 patients requiring vascular reconstruction during a robotic pancreaticoduodenectomy (11). In addition to demonstrating efficacy in using the robotic system for vascular reconstruction, this review highlights that there is an inverse relationship between experience (i.e., the number of cases performed) and operative time (11).
Part II: endovascular-first procedures
IVC filter removal
One of the most widely written about applications of robot-assisted procedures within the field of vascular surgery is in the removal of IVC filters. Most frequently, IVC filters may be removed via an endovascular approach. However, there are a certain number of instances in which the filters are not amenable to endovascular recovery, and alternative options must be explored to facilitate safe removal. One common example when an endovascular approach is not safe, is when the filter struts fracture and become embedded within the wall of the IVC. This leads to a particularly dangerous situation in which any attempt for endovascular recovery may result in an injury to the IVC and uncontrollable hemorrhage. In 2020, Lin et al. published a case report detailing the successful recovery of an IVC filter utilizing robot technology (12). In this case report, Dr. Lin utilized a Da Vinci robot to remove an IVC filter that became malpositioned with a fractured strut protruding outside of the vena cava (12). The filter was able to be successfully removed with exposure of the IVC utilizing robot technology. The procedure lasted 189 minutes with an estimated blood loss of 200 mL. The patient was discharged home on postoperative day 2. Since this case report, there have been multiple case series demonstrating the use of robot technology in assisting with exposure of the IVC for filter removal.
The use of the robot in common angiograms
In addition to utilizing robot technology to assist in performing typically open vascular repairs and reconstructions, there is also a new path being forged in utilizing the robot to assist in wire manipulation and vessel targeting during common endovascular interventions. Over the last 100 years, endovascular interventions have rapidly grown within the field of vascular surgery and offer benefits such as fewer complications, quicker recovery, shorter hospital length of stay and avoidance of major abdominal procedures (13). While endovascular interventions have transformed the field of vascular surgery, there are a number of challenges and drawbacks. One of the challenges in an endovascular approach lies in one’s ability to correctly target the appropriate vessel, especially in areas that require complex anatomic navigation. The second challenge is the risk of distal embolization, and the third is in the exposure of physicians and support staff to high levels of radiation (13).
These limitations have led to the development of novel robotic remote-controlled catheter navigation systems which offer control either via an electromechanical- or magnetic-based system. The electromechanical-based systems include the Amigo, Sensei, Magellan, and CorPath vascular robotic systems, while the magnetic-based navigation system includes the Niobe robotic system.
This technology was first utilized for robot-assisted percutaneous coronary intervention and described by Cruddas et al. (13). These robotic systems allow for manipulation of guidewires and catheters to rotate in 30-degree increments combined with 1 mm advancements and retractions to allow for precise positioning within complex anatomy (13). Multiple case series have been conducted from 2013 to 2021 evaluating these systems for treating carotid arteries, endovascular repair (EVAR) gate cannulation, and even angioplasties of the superficial femoral artery (13). In 2018, the CorPath system was approved for use in the treatment of peripheral arterial disease.
In 2015, Cochennec et al. utilized the Magellan robotic system in 37 attempts for complex endovascular aortic procedures (14). They were able to successfully cannulate the intended vessel in less than 15 minutes in 81% (30/37) of the attempts. In the remaining seven cases, they were eventually able to cannulate with the robot system via branches and chimney stents. This study highlighted that complex endovascular maneuvers could be successfully and quickly performed with the robotic systems (14).
In 2016, the Robotic-Assisted Peripheral Intervention for peripheral arterial Disease (RAPID) study was a prospective single-arm, single-center and nonrandomized trial that evaluated 20 patients undergoing robotic peripheral vascular interventions (15). The majority of the patients in this study underwent intervention in the superficial femoral artery. The study reported a technical success rate of 96.6% with no adverse clinical events. In addition, the study reported a total fluoroscopy time of 7.1±3.2 minutes which compared favorably to a traditional endovascular approach with a mean of 13–15 minutes in similar patient cohorts (16).
Overall, the greatest benefit of robot-assisted endovascular procedures is in the significant reduction in radiation exposure (13). This reduction in radiation was evaluated in the Percutaneous Robotically-Enhanced Coronary Intervention (PRECISE) study. This study found that radiation exposure of the primary operator was 95.2% lower compared to levels found during traditional procedures (17). In addition to decreasing radiation exposure, there are also technologies being incorporated to utilize 3D visualization to improve navigation in hopes to reduce the use of radiographic contrast (13).
While there are certainly advantages in the utilization of robot technology for assistance in endovascular procedures, there are drawbacks as well. One primary drawback is the sheath size that is required for access. For example, the Magellan system requires a 14-Fr sheath, whereas a traditional endovascular approach may be performed with a 5- or 6-Fr Sheath (13). A second drawback in the use of robot technology for endovascular procedures is the loss of haptic feedback. Oftentimes an experienced vascular surgeon will be able to ascertain critical information about the anatomy of a particular lesion by precise manipulation of a wire. One example of this haptic feedback is in the ability for an experienced surgeon to estimate the age of a thrombus or clot based upon its pliability and ease at which the wire passes through. A surgeon’s ability to ascertain this critical knowledge and detailed anatomy of the patient’s vessels may allow one to avoid the unintended consequences of wire manipulation, such as in distal embolization and in the creation of dissection planes (13). A final drawback in the use of robot technology, is that while the robot-system offers great advantages in allowing one to gain access to the intended location within the vessel, the clinician must oftentimes still physically scrub into the procedure to inflate the balloon, or deploy the stent.
Barriers to the incorporation of robot technology
While there appears to be several advantages to the use of robot technology within the field of vascular surgery, there remain a few barriers that stall its widespread use. The first is a purely economic one, as the upfront cost for each robot system is approximately $1.5 million dollars, with an additional service cost of $112,000 per year (1). In order for robot technology to continue to advance the field of vascular surgery, institutions must be willing to make sizable economic commitments.
A second disadvantage lies in the steep learning curve that one must overcome to become an effective and safe operator of the robotic system. While it is true that the learning curve is present for all surgeons who wish to incorporate robotic technology within their practice, an added difficulty specifically within the field of vascular surgery is in the rising popularity of the integrated vascular surgery residency programs. Many surgeons who graduate from a traditional general surgery residency program have already undergone intense training with both laparoscopic and robotic instruments. Most of these residents have already logged hundreds of hours of direct training using these tools in procedures ranging from cholecystectomies to colon resections. Residents who progress through this general training, and then pursue vascular fellowship will have a broad minimally invasive base from which to further develop their skills. In contrast, residents of an integrated vascular surgery residency program may not have experienced the same exposure to minimally invasive techniques. For that reason, this later subgroup of physicians will likely require additional training that represents a large opportunity cost to both the individual and the institution he or she is associated with.
The final barrier to the widespread incorporation of robot technology, is the lack of randomized controlled trials specifically comparing traditional methods and robotic-assisted ones. The majority of the articles examined for this narrative review were retrospective in nature, and further research is crucial to prove there is a benefit in widespread adaptation of this technology in the field of vascular surgery.
Conclusions
Overall, this review highlights the multitude of uses for minimally invasive robot-assisted interventions within the field of vascular surgery, ranging from complex aortofemoral reconstructions, management of venous pathology, venous reconstruction in oncologic resection, complex IVC filter removal and even in improving endovascular-based therapies. While the current trajectory shows that robot technology will likely continue to advance the field of vascular surgery, there remain several large hurdles that must be overcome prior to its widespread use for the common vascular surgeon.
Acknowledgments
The authors would like to formally thank the senior members of our group for their unwavering support and commitment throughout this project.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by editorial office, Annals of Laparoscopic and Endoscopic Surgery for the series “Latest MIS Approaches and Data”. The article has undergone external peer review.
Reporting Checklist: The authors have completed the Narrative Review reporting checklist. Available at https://ales.amegroups.com/article/view/10.21037/ales-23-24/rc
Peer Review File: Available at https://ales.amegroups.com/article/view/10.21037/ales-23-24/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://ales.amegroups.com/article/view/10.21037/ales-23-24/coif). The series “Latest MIS Approaches and Data” was commissioned by the editorial office without any funding or sponsorship. GF served as the unpaid Guest Editor of the series and serves as an unpaid editorial board member of Annals of Laparoscopic and Endoscopic Surgery from April 2023 to March 2025. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Püschel A, Schafmayer C, Groß J. Robot-assisted techniques in vascular and endovascular surgery. Langenbecks Arch Surg 2022;407:1789-95. [Crossref] [PubMed]
- Dion YM, Kathouda N, Rouleau C, et al. Laparoscopy-assisted aortobifemoral bypass. Surg Laparosc Endosc 1993;3:425-9. [PubMed]
- Norgren L, Hiatt WR, Dormandy JA, et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg 2007;45 Suppl S:S5-67.
- Wisselink W, Cuesta MA, Gracia C, et al. Robot-assisted laparoscopic aortobifemoral bypass for aortoiliac occlusive disease: a report of two cases. J Vasc Surg 2002;36:1079-82. [Crossref] [PubMed]
- Štádler P, Dvořáček L, Vitásek P, et al. Robot assisted Aortic and Non-aortic Vascular Operations. Eur J Vasc Endovasc Surg 2016;52:22-8. [Crossref] [PubMed]
- Coggia M, Javerliat I, Di Centa I, et al. Total laparoscopic versus conventional abdominal aortic aneurysm repair: a case-control study. J Vasc Surg 2005;42:906-11. [Crossref] [PubMed]
- Gharagozloo F, Meyer M, Tempesta B, et al. Robotic transthoracic first-rib resection for Paget-Schroetter syndrome. Eur J Cardiothorac Surg 2019;55:434-9. [Crossref] [PubMed]
- Wang B, Huang Q, Liu K, et al. Robot-assisted Level III-IV Inferior Vena Cava Thrombectomy: Initial Series with Step-by-step Procedures and 1-yr Outcomes. Eur Urol 2020;78:77-86. [Crossref] [PubMed]
- Quencer KB, Friedman T, Sheth R, et al. Tumor thrombus: incidence, imaging, prognosis and treatment. Cardiovasc Diagn Ther 2017;7:S165-77. [Crossref] [PubMed]
- Yamamoto M, Omori T, Shinno N, et al. Robotic total gastrectomy with thrombectomy and portal vein reconstruction for gastric cancer and portal vein tumor thrombus. World J Surg Oncol 2022;20:36. [Crossref] [PubMed]
- Beane JD, Zenati M, Hamad A, et al. Robotic pancreatoduodenectomy with vascular resection: Outcomes and learning curve. Surgery 2019;166:8-14. [Crossref] [PubMed]
- Lin JC, Patel A, Rogers CG. Robot-assisted removal of inferior vena cava filter. J Vasc Surg Cases Innov Tech 2020;6:311-2. [Crossref] [PubMed]
- Cruddas L, Martin G, Riga C. Robotic endovascular surgery: current and future practice. Semin Vasc Surg 2021;34:233-40. [Crossref] [PubMed]
- Cochennec F, Kobeiter H, Gohel M, et al. Feasibility and safety of renal and visceral target vessel cannulation using robotically steerable catheters during complex endovascular aortic procedures. J Endovasc Ther 2015;22:187-93. [Crossref] [PubMed]
- Mahmud E, Schmid F, Kalmar P, et al. Feasibility and Safety of Robotic Peripheral Vascular Interventions: Results of the RAPID Trial. JACC Cardiovasc Interv 2016;9:2058-64. [Crossref] [PubMed]
- Mahmud E, Schmid F, Kalmar P, et al. Robotic Peripheral Vascular Intervention With Drug-Coated Balloons is Feasible and Reduces Operator Radiation Exposure: Results of the Robotic-Assisted Peripheral Intervention for Peripheral Artery Disease (RAPID) Study II. J Invasive Cardiol 2020;32:380-4. [PubMed]
- Weisz G, Metzger DC, Caputo RP, et al. Safety and feasibility of robotic percutaneous coronary intervention: PRECISE (Percutaneous Robotically-Enhanced Coronary Intervention) Study. J Am Coll Cardiol 2013;61:1596-600. [Crossref] [PubMed]
Cite this article as: Huber MA, Robbins JM, Sebastian SM, Vu AH, Ferzli G, Schutzer R, Hingorani A. The robotic approach for vascular and endovascular procedures: a narrative review. Ann Laparosc Endosc Surg 2023;8:33.